Memantine Hydrochloride Tablets IP
Memantine Hydrochloride Tablets IP Specification
- Indication
- Moderate to severe Alzheimers disease, Dementia associated with Alzheimers
- Origin of Medicine
- India
- Brand Name
- Memantine Hydrochloride Tablets IP
- Life Span
- 24 Months
- Salt Composition
- Memantine Hydrochloride
- Pacakaging (Quantity Per Box)
- 30 Tablets
- Packaging Type
- Blister pack (Box)
- Dosage Form
- Oral tablet
- Drug Type
- Allopathic
- Ingredients
- Memantine Hydrochloride
- Physical Form
- Tablets
- Function
- NMDA receptor antagonist for neuroprotection
- Recommended For
- Treatment of Alzheimers disease and moderate to severe dementia
- Dosage
- As directed by physician, typically starting at 5 mg once daily, titrated up to 20 mg per day
- Dosage Guidelines
- Follow physicians instructions; do not self-medicate
- Suitable For
- Adults, Geriatric Patients
- Quantity
- 30 tablets per box
- Storage Instructions
- Store below 30C in a dry place, protect from light and moisture
- Side Effects
- Dizziness, headache, confusion, constipation
- Regulatory Status
- Scheduled H drug in India
- Contraindications
- Known hypersensitivity to Memantine or any excipients
- Prescription Status
- Prescription only (Rx)
- Administration Route
- Oral
- Color
- White or off-white
- Compliance Standard
- IP (Indian Pharmacopoeia)
- Marketed by
- As per packaging details
- Safety Information
- Not advised for use in children; consult doctor for renal/hepatic impairment
- Shape
- Round, film-coated tablet
- Batch Number
- As printed on blister pack
- MRP
- As per retail packaging
- Strength
- 5 mg or 10 mg per tablet (confirm pack label)
Memantine Hydrochloride Tablets IP Trade Information
- Minimum Order Quantity
- 100 Packs
- Payment Terms
- Cash in Advance (CID)
- Supply Ability
- 5000 Packs Per Month
- Delivery Time
- 10 Days
- Main Domestic Market
- All India
About Memantine Hydrochloride Tablets IP
Surface Application, Special Features & Proper Usage Directions
Memantine Hydrochloride Tablets IP deliver precise neuroprotection benefits on the neural application surface, ensuring masterful support for cognitive function in dementia patients. This pharmaceutical stands out with its easy-to-administer, round, film-coated tablet design, optimized for adult and geriatric use. For best outcomes, follow your physician's directions precisely-begin with 5 mg once daily, titrating up as advised. Do not self-medicate. Its resplendent efficacy makes it a favoured choice for advanced Alzheimer's care.
FOB Port, Export Markets & Certifications-A Trusted Global Solution
Memantine Hydrochloride Tablets IP are handed over via major Indian FOB ports upon confirmed purchase order, ensuring smooth international trade. These acclaimed tablets are widely handed over to markets across Asia, Africa, and the Middle East, making them a renowned global solution. Certified under the Indian Pharmacopoeia (IP) and adhering to Scheduled H regulatory standards, every consignment is traced by batch number for assured quality. Purchase with confidence-reliability and compliance are guaranteed upon every handover.
FAQ's of Memantine Hydrochloride Tablets IP:
Q: How should Memantine Hydrochloride Tablets IP be administered?
A: Memantine Hydrochloride Tablets IP should be taken orally, starting typically at 5 mg once daily, with the dose titrated up to a maximum of 20 mg per day as directed by your physician. Always follow your doctor's instructions and never self-medicate.Q: What conditions are Memantine Hydrochloride Tablets IP recommended for?
A: These tablets are recommended for the treatment of moderate to severe Alzheimer's disease and dementia associated with Alzheimer's. They function as NMDA receptor antagonists to support cognitive health and neuroprotection.Q: When should I avoid taking Memantine Hydrochloride Tablets IP?
A: Avoid using this medication if you have a known hypersensitivity to Memantine or any tablet excipients. It's also not advised for children and should be used cautiously in patients with renal or hepatic impairment-always consult your healthcare provider first.Q: What are the potential side effects of using Memantine Hydrochloride Tablets IP?
A: Some individuals may experience dizziness, headache, confusion, or constipation while taking Memantine Hydrochloride Tablets IP. Speak to your physician if you notice any concerning symptoms.Q: Where are Memantine Hydrochloride Tablets IP manufactured and exported?
A: These tablets are produced in India, complying with IP standards, and exported to multiple regions, including Asia, Africa, and the Middle East, via major Indian FOB ports as part of a secure and timely supply process.Q: What makes this product a hot-selling choice among traders and suppliers?
A: Memantine Hydrochloride Tablets IP are highly sought after due to their resplendent effectiveness, adherence to stringent regulatory standards, and robust global distribution network. Their proven efficacy in Alzheimer's care makes them favored among healthcare professionals.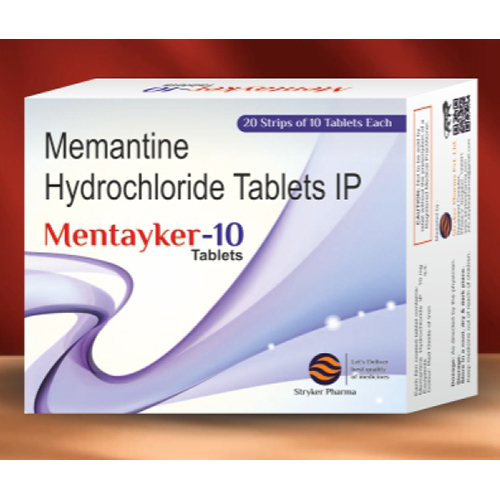

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Neurology Medicine Category
Levetiracetam Tablets IP
Minimum Order Quantity : 100 Packs
Dosage Form : Oral tablet
Storage Instructions : Store in a cool, dry place below 30C, protect from moisture and light
Packaging Type : Blister pack
Suitable For : Other, Adults and children as prescribed
Recommended For : Used in the treatment of epilepsy and seizures
Levetiracetam Tablets IP
Minimum Order Quantity : 100 Packs
Dosage Form : Tablet
Storage Instructions : Store in a cool, dry & dark place below 30C. Protect from light & moisture.
Packaging Type : Blister Strips
Suitable For : Other, Adults, Children
Recommended For : Adults and children with epilepsy
Levetiracetam Tablets IP
Minimum Order Quantity : 100 Packs
Dosage Form : Oral tablet
Storage Instructions : Store in a cool, dry place below 30C, protect from light and moisture
Packaging Type : AluAlu or Blister pack
Suitable For : Other, Adults and children as directed
Recommended For : Epilepsy, seizure disorders
10 MG GDonepezil Tablets IP
Minimum Order Quantity : 100 Packs
Dosage Form : Oral tablets
Storage Instructions : Store in a cool, dry place below 30C. Protect from light and moisture.
Packaging Type : Blister strip
Suitable For : Other, Adults and elderly patients
Recommended For : Alzheimers disease and dementia in elderly

 Send Inquiry
Send Inquiry Send Inquiry
Send Inquiry